About paper
Czech originalHeat pack
Abstract
It is possible for some substances to be kept in liquid state at temperatures and pressures below the triple point under certain circumstances. Crystallization in such state releases the internal energy in the form of heat. Simple measurements concerning this phenomenon can be demonstrated with a heat pack (firebag).
Description of Operation
The heat pack was covered by patent number 4,077,390 in California in 1978. In the Czech Republic it can be bought in sport shops or pharmacies today. The heat packs used for our measurements were made in Germany. The information on heat packs which are sold under the trademark “Firebag” states that the working fluid is sodium acetate (Figure 1). The pack is made of plastic bag filled with colorless fluid in which there floats an oval tinny disc.

Fig. 1 Heat pack “Firebag” – front and back
The heat pack is activated by bending the disc through the pack and releasing it. Rapid crystallization takes place in the pack (if it is not out of order). The solid phase appears at one point of the rim of the disc and it fills the whole volume of the pack in approximately 10 s. The heat pack gets surprisingly hot during crystallization and it is capable to provide heat for example to chilled hands for quite a long period. (The actual duration of heating depends on the temperature of the outside of the pack.) The interior of the pack is very hard and its crystalline structure is clearly visible after cooling down to room temperature.
The heat pack can be warmed in hot water and the crystals melt little by little to the original state which is colorless liquid. The producer states that it is possible to perform about 500 cycles.
Motivational Experiment
If we have a sufficient number of heat packs, we can hand them out to the students so that everyone can try out starting the crystallization process. The phenomenon is so nice and surprising that it will motivate the students to examine the process further.
The following questions arise:
1. What temperature does the heat pack reach during crystallization?
2. How much heat can it give until it is cooled to the original temperature?
3. Why did the heat pack heat up?
4. Why did crystallization take place?
The students can suggest themselves what is necessary to be measured and with what methods to answer the question.
Measurements and Calculations
1. The temperature of the heat pack can be measured with a usual liquid or digital thermometer. A hot heat pack is not completely tough and can be bent and wrapped around the thermometer. The students can estimate the temperature before the measurement. Repeated measurements yielded a temperature of about 52 °C.
2. The amount of heat that the heat pack can give to the surroundings can be measured with a calorimeter. We can measure the specific heat capacity of the crystalline phase of sodium acetate by inserting the heat pack into cold water in the calorimeter. From specific heat we can calculate the heat that is given by the heat pack during cooling from 52 °C to room temperature.
We give the measured data in Table 1.
The initial temperature of the pack in liquid state was t = 24.5 °C and the temperature of the heat pack just after crystallization was t* = 52 °C.
The mass of the inner vessel of the calorimeter is m2 = 215 g. It is made of aluminium that has (according to the tables) specific heat capacity of c2 = 896 J·kg·K–1. The calorimeter was filled with water (specific heat capacity acc. to tables c1 = 4 180 J·kg-1·K-1). The mass of the heat pack (including the plastic bag and the tinny disc) is m'. We measured the mass of the plastic bag and the disc of a gashed heat pack – they weigh approximately 11 g together. The mass m of sodium acetate solution was calculated as the difference of the whole pack’s mass and these 11 g (assuming that the pack and the disc weigh approximately the same in all heat packs).
We put the heat pack into the calorimeter when we initiated crystallization. We stirred the water in calorimeter from time to time and waited until its temperature stabilized at a value t3. If we assume that the heat is transferred only between the heat pack, water, and the inner vessel of the calorimeter, we can calculate the heat Q1 that was transferred from the heat pack to the system until it reached thermodynamical equilibrium
\[Q_1 = c_1 m_1 (t_3 – t_1) + c_2 m_2 (t_3 – t_1) \]Then we calculate the specific heat capacity of crystalline phase of sodium acetate in the heat pack using the formula Q1 = c m (t* – t3):
\[ c = \frac{Q_1}{m(t^* - t_3)}. \]Using c we can calculate the heat Q2 that the heat pack would give to cool itself further from temperature t3 to the initial temperature before crystallization t: \[Q_3 = c m (t_3 – t) .\]
Then we find Q = Q1 + Q2.
Table 1 Measured Data
| t1/°C |
m1/g |
m2/g |
t/°C |
t3/°C |
m/g |
t*/°C |
Q1/J |
c/J kg-1 K-1 |
Q2/J |
Q/kJ |
| 24 |
334.1 |
215 |
24.5 |
33 |
118 |
52 |
14303 |
6379 |
6399 |
20.7 |
| 25.5 |
386.4 |
215 |
24.5 |
34 |
120 |
52 |
15366 |
7114 |
8110 |
23.5 |
| 25.5 |
396.4 |
215 |
24.5 |
33.5 |
114 |
52 |
14797 |
7016 |
7198 |
22.0 |
| t1 |
initial temperature of water |
| m1 |
mass of water |
| m2 |
mass of the inner vessel of the calorimeter |
| t |
initial temperature of the heat pack |
| t3 |
temperature in the calorimeter in thermodynamic equilibrium |
| m |
mass of sodium acetate |
| t* |
temperature of heat pack after crystallization |
| Q1 |
heat accepted by water and calorimeter |
| c |
specific heat capacity of crystalline sodium acetate |
| Q2 |
heat that could be given by the heat pack subsequently |
| Q |
the total heat produced by the heat pack |
The measured average value of specific heat capacity of crystalline phase of sodium acetate solution in the heat pack is c = 6 800 J kg–1 K–1, the average heat that can be given by the heat pack is Q = 22 kJ.
If we introduce caloric capacity of the sodium acetate solution in the heat pack as \(\frac{Q}{m}\), we get an average value of 188 kJ kg–1.
Where Does the Heat of the Pack Come From?
The explanation of the processes that take place in the heat pack is not simple. The patent description speaks about a supercoolable aqueous solution. There can be found a lot of articles in foreign languages that explain the phenomenon in various ways on the Internet. We read terms such as supercooled liquid, supersaturated solution or undercooled melt. The liberated heat is called heat of phase transition. Initiation of crystallization is connected to releasing of crystallization nuclei from the micro fissures in the disc or possibly to mere bending of the tinny disc.
Let us see, what high school students do know. In physics they learned that cooling of a liquid leads to its solidification. If the liquid was formed by melting of a crystalline material, crystallization nuclei occur first when the liquid is cooled at its melting point etc. Some students do know the appendix written in [1]: When cooling a pure substance, it often happens that crystallization nuclei emerge at temperature lower than the melting point of the substance. The liquid at temperature below its freezing point is referred to as supercooled. Sodium thiosulphate that has melting point at 48 °C can be liquid at 20 °C. If we throw some crystals of sodium thiosulphate in such supercooled liquid, it transits rapidly to the solid state and its temperature grows to the melting point.
How to explain to the students where did the heat come from? If we do not want to use terms of chemists, we should speak about transfer of potential energy to kinetic energy. Plainly we can say that the crystalline phase has stronger bonds than liquid phase. Stronger (or closer) bonds mean lower potential energy. Because the law of conservation of energy is valid also in the world of atoms, molecules and ions, the kinetic energy of their oscillation must grow. This is manifested by warming of the heat pack.
We see a problem in understanding the concept of potential energy of two adjacent particles. Simplified explanation should use the concept of gravitational potential energy (Ep = mgh) that is best comprehended by high school students. If the distance h between the body and the Earth decreases, the potential energy Ep decreases, too. If a body is falling to the ground, its potential energy decreases and its kinetic motional energy increases. This analogy is a bit imprecise but acceptable for 2nd year high school students.
Thermonuclear Fusion
Until now we have been thinking only about thermodynamics but the heat pack can also be useful when explaining thermonuclear synthesis that also does liberate heat. The processes are technically the same. In the beginning we have to add some energy to get the atomic nuclei sufficiently close together to put them under a stronger influence of nuclear forces. As soon as new stronger bonds begin to arise between the nucleons, the potential energy of the boundary forces decreases and the kinetic energy of oscillation increases. That induces rapid warming of the matter.
Older students should be capable to understand the key differences between the heat pack and thermonuclear fusion. It is the potential energy of electrostatic forces that warms up the heat pack (the energy comes from electron cloud), in case of thermonuclear fusion it is the potential energy of nuclear forces that warms up the matter (the energy is liberated from the nucleus). The amount of released energy is very different, of course.
Electric Potential Energy
The term of electric potential energy can be specified during optional physics lessons. We will derive potential energy of two opposite charges Q1 a Q1 between which there is distance r (Figure 2).
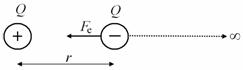
Fig. 2 Scheme of electrostatic force between two opposite charges
We must begin with definition of potential energy.
1. If the distance between the charges is infinite, their mutual potential energy is zero.
2. For a finite distance r, the electric potential energy is equal to the work that the electric field would have to do to move the charges to the distance of zero potential energy.
Because Coulomb’s electrostatic force Fe is inversely proportional to the square of the distance x between the charges \[ F_e = \frac{1}{4\pi \epsilon_0} \frac{|Q_1| \, |Q_2|}{x^2} ,\] we have to calculate the work by integration. We must take into account that the angel α between the direction of charge’s movement and the electrostatic force is 180°.
\[ E_p = W = \int^\infty_r F_e \mathrm{d}x \cos \alpha = \int^\infty_r \frac{1}{4\pi \epsilon_0} \frac{|Q_1| \, |Q_2|}{x^2} \mathrm{d}x (-1) = \] \[ = - \frac{|Q_1| \, |Q_2|}{4\pi \epsilon_0} \left[ - \frac{1}{r} \right]^\infty_r = - \frac{|Q_1| \, |Q_2|}{4\pi \epsilon_0 r}. \]The definition yields logically that the potential energy of two opposite charges at finite distance is negative. The resultant formula can be used in an exercise.
Exercise: Let there be two opposite elementary point charges in the distance of 0.3 nm. How much will their kinetic energy increase if they move to a distance of 0.2 nm?
\[|Q_1|=|Q_1|=e=1.6 \cdot 10^{-19}\,\mathrm{C}, r_1 = 0.3\,\mathrm{nm}, r_2=0.2\,\mathrm{nm} \]Solution: The law of conservation of energy is in force:
\[ E_{p1} + E_{k1} = E_{p2} + E_{k2} \] \[ - \frac{e^2}{4\pi \epsilon_0 r_1} + E_{k1} = - \frac{e^2}{4\pi \epsilon_0 r_2} + E_{k2} \] \[ \Delta E_k = E_{k2} - E_{k1} = \frac{e^2}{4\pi \epsilon_0 r_2} - \frac{e^2}{4\pi \epsilon_0 r_1} = \frac{e^2}{4\pi \epsilon_0 } \left( \frac{1}{r_2} - \frac{1}{r_1} \right) \] \[\Delta E_k = 3.83\cdot 10^{-19}\,\mathrm{J} = 2.4\,\,\mathrm{eV}\]The fact that potential energy of two opposite charges is negative is not given in the textbook [1] among models of liquid/gaseous/solid states. On the contrary the textbook says that the potential energy of boundary forces is larger than the kinetic energy of the vibrational movement in solid state. This proposition is in contradiction with its negative value.
The boundary nuclear energy Ej and boundary energy associated to 1 nucleon εj in textbook [2] should also be introduced as negative. It would be easier for the teacher to work with the law of conservation of energy again. In such case the mass defect would also need to be introduced as a negative number. The author uses this convention in his lessons of physics.
The following exercise will be solved similarly.
Exercise: Count the energy that is liberated during thermonuclear synthesis of deuterium and tritium to helium4He. The boundary energies associated with one nucleon are
\[ \epsilon_j\left(\mathrm{^2_1H}\right) = -1.11\,\mathrm{MeV}, \epsilon_j\left(\mathrm{^3_1H}\right) = -2.83\,\mathrm{MeV}, \epsilon_j\left(\mathrm{^4_2He}\right) = -7.07\,\mathrm{MeV}. \]Solution: First we write the equation of thermonuclear synthesis
\[ \mathrm{^2_1H + ^3_1H \rightarrow ^4_2He + ^1_0n } \]The law of conservation of energy is in force again so the sum of nuclear (potential) energy and kinetic energy is conserved.
\[ E_{j1} + E_{k1} = E_{j2} + E_{k2}\] \[ \Delta E_k = E_{k2} - E_{k1} = E_{j1} + E_{j2} = 2 \epsilon_j\left(\mathrm{^2_1H}\right) + 3 \epsilon_j\left(\mathrm{^3_1H}\right) - 4 \epsilon_j\left(\mathrm{^4_2He}\right) \]…the neutron has zero boundary energy
\[ \Delta E_k = 2\cdot (-1.11\,\mathrm{MeV}) + 3 \cdot (-2.83\,\mathrm{MeV}) -4 \cdot (-7.07\,\mathrm{MeV}) = 17.57\,\mathrm{MeV} \]The change of kinetic energy is positive which means that the non-boundary energy of the system has increased and the matter has warmed up.
Mechanism of the Processes in the Heat Pack
What is going on in the heat pack after bending the disc? The following paragraphs stand on articles we have read and on our own experimenting.
Let us examine the properties of sodium acetate trihydrate CH3COONa·3H2O. This substance has density of 1.45 g·cm-3 in solid state. It crystallizes in monoclinic crystal system. It creates colorless crystals in which there are water dipoles intercalated between the ions. When heated to 58 °C, the crystal lattice gives out the molecules of water in which the sodium acetate subsequently dissolves. The substance melts and is dissociated into ions CH3COO- and Na+.
Yet cooling of the solution below 58 °C does not lead to crystallization. The solution cools down to room temperature and exists as supercooled supersaturated solution.
So we can state that there is a supercooled solution in the pack. Bending of the disc induces crystallization that liberates heat as described above.
The last thing remaining to be explained is how does bending of the disc induce crystallization. The thought that there are microcrystals of sodium acetate (working as crystallization nuclei) released from the fissures in the disc is disconfirmed by the following experiment: We melted only a portion of the crystals in the heat pack in warm water and let it cool down (Figure 3). Although there are clusters of crystals floating in the liquid, crystallization does not occur spontaneously.

Fig. 3 Cold heat pack with some unmelted crystals of sodium acetate
If we bend the disc in the heat pack in this state, crystallization passes off normally. We managed to induce crystallization (in some packs) also by pressing the crystalline clusters against each other!
We tested the hypothesis that bending of the disc sends out a shock wave that induces crystallization. Yet inducing of a shock wave in the heat pack by other means such as striking the pack with a blunt object or throwing it against a solid obstacle does not start crystallization. Some discs do not even perform the typical “click” that would send out a wave.
Conscientious watching of the beginning of the crystallization shows that a centre of crystallization occurs in one of the fissures (Figure 4) that are pressed in the disc no sooner than we bent the disc and released it. The crystallization then propagates to all parts of the heat pack.
It seems that the fissures in the disc are the best places for the crystallization induced by the pressure of our fingers to occur.

Fig. 4 The tinny disc that starts crystallization in the heat pack
The idea of necessity of certain pressure is confirmed by the experiment with floating clusters of crystals.
There is one more interesting phenomenon. Some packs lose the ability to crystallize but if they were pierced with a metallic spike, crystallization started from the point where the spike got into the solution.
Sodium Acetate Column
Finally let us show an experiment with supercooled solution of sodium acetate that is not in the heat pack. We melt crystalline sodium acetate in a beaker and let it cool down to room temperature. We put some sodium acetate crystals on a small plate and we slowly pour cold liquid solution from the beaker on them. The solution crystallizes on the plate immediately and we can grow a candle-like column of sodium acetate on the plate (see Figure 5). The whole column is heated during crystallization, of course.

Fig. 5 Column created by pouring of supercooled solution on some crystals of sodium acetate
Conclusion
The article gives information about the function of a heat pack and tries to explain the processes in the heat pack from the physical point of view. It describes the use of heat packs as motivational qualitative experiment in physics lessons as well as quantitative measurement with the heat pack in thermodynamical practical training. The concept of electric potential energy of two charges is introduced beyond the usual high school curriculum. Exercises on calculating the energy liberated during crystallization and during thermonuclear synthesis are introduced in this article, too.
References
[1] K. Baruška, E. Svoboda, „Molekulová fyzika a termika“, (1993)
[2] I. Štoll, „Fyzika mikrosvěta“, (1993)
[3] Patent No. 4,077,390, Reusable heat pack containing supercooled solution and means for activating same, United States Patent and Trademark Office, http://patft.uspto.gov/
[4] Sodium Acetate, Wikipedia, http://en.wikipedia.org/wiki/Sodium_acetate
[5] Prof. Blumes Bildungsserver für Chemie, Tipp des Monats Januar 1999, Wärmekissen: Schnelle Wärme aus Kristallen, http://dc2.uni-bielefeld.de/dc2/index.html